- Adult body size: 0.1–1.5 millimeters
- Number of legs: 8
- First discovered: 1773
- Estimated history on Earth: 600 million years
- Easily found in: lichens, mosses, dunes, beaches, ponds
- Shrugs off extremes of: radiation, pressure, temperature…
Since she first learned about them in day care, my five-year-old has been mesmerized by microbes. She swats them out of the air, she stomps her feet to crush them; she is ruthless. And one day, when she told me that she wanted to see some microbes, I knew just the man to call.
A week later we found ourselves in Bob Goldstein’s biology lab, peering through his microscope at a curious little critter that looked like an overstuffed couch with eight legs, tiny claw-like toes, a little round head, and two beady eyespots. It was bumbling through the water, munching on specks of something too small to see. Until that morning, it had been living in Goldstein’s back yard on a bit of moss. It’s a tardigrade, Goldstein explained. (Tardi-what?) They’re also called water bears, he added. I was hooked.
Tardigrades — the name comes from the Latin for “slow walker” — are harmless to humans. But they just might be the most oddball animals on the planet. They can live almost anywhere, from the highest mountains to the deepest seas, at the poles or the equator. They like moisture, but when they don’t have enough they can enter a kind of dry hibernation called the tun state. Once in that state, they are impressively hard to kill. They can survive doses of x-ray radiation hundreds of times stronger than you or I could live through. They can withstand extreme temperatures — scientists have heated them to 300 degrees Fahrenheit and cooled them to within a degree or two of absolute zero, the temperature at which all molecular motion stops. Pressure extremes don’t faze them, either. They have survived both the vacuum and the solar radiation of open space, something no other animal can do. In fact, it doesn’t much matter what you try to do to them: “Just put them back into some water,” Goldstein says, “and they walk away to tell about it.” No one fully understands how tardigrades pull this off.
But what are they, I wondered? Are they like plankton? Some kind of microscopic insect? No, Goldstein explained. They’re tardigrades. On the tree of life, they have their own phylum. We humans share a phylum with sea squirts, crocodiles, and about a hundred thousand other animals. Tardigrades hoe their own row.
There are around one thousand known species of the little guys. “But there are probably several thousand species out there,” Goldstein says. “Most tardigrades you’d find are new species.” (See related story: “Every Living Thing.”)
Goldstein is a developmental biologist who’s also interested in evolution, in how all the different organisms on Earth got here. The journey from fertilized egg to fully formed animal is complicated and not well understood. “We know a bit about how development works in specific organisms,” Goldstein says. Fiddle with the right fruit fly genes, for example, and you can end up with flies that have unusual eye coloring or longer wings. Broadly, that’s how evolution works: small changes in DNA, over time, form new species. But how?
Two organisms that outwardly seem nothing alike can have a lot of genetic overlap. Your limbs and a fruit fly’s legs are built by the same genes. Sean Carroll, a leading evolutionary biologist, compares development to choreography. “You’ve got the same dancers,” he told Discover magazine, “but the ballet is based on different cues.”
Goldstein says that if you look at images of bat embryos taken at different developmental stages, “the embryos look like a mouse, look like a mouse, look like a mouse… and all of a sudden you see that these giant wings have developed where there would have been mouse legs otherwise.” How did we get from mouse to bat? Goldstein’s work, he says, is about understanding the trajectories evolution can and can’t take.
When he came to Carolina in 1999, Goldstein was looking for an organism he could use to study evolutionary development. He settled on tardigrades. They don’t have too many genes. He can grow them in the lab. They develop reasonably quickly. Their embryos are small and clear, so he can see what’s going on inside. And their branch on the tree of life happens to be right next to the branches of two of science’s most-studied organisms: the nematode C. elegans and the fruit fly Drosophila (See related story: “Fly Club.”) “The fact that tardigrades are closely related to these other organisms means they probably have roughly the same genomes,” Goldstein says. “And that means we can make one-to-one comparisons more often than we could with a distant relative.” Those comparisons may help scientists figure out how very similar genomes can produce very different animals.
Goldstein and his lab are pretty much the only scientists anywhere who study evolutionary development in tardigrades. He wants to find a way to mess with a tardigrade gene to see what happens when it’s not working properly. Jenny Tenlen, a postdoc in Goldstein’s lab, is using RNA interference to do that, and Goldstein says it seems to be working. “This was one of the big hurdles: if we couldn’t find any method to disrupt gene function, then we weren’t going very far at all. I went into this project thinking I’d only continue for as long as it gave us green lights,” he says, “and it’s just given us one green light after another.”
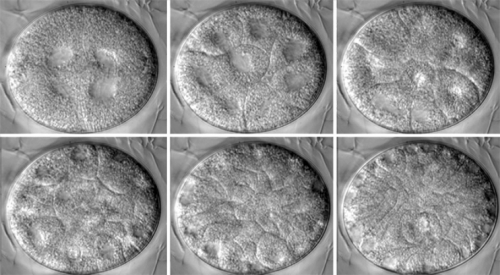
Goldstein is a gadget guy — he has three or four Roomba robotic vacuum cleaners, one of which cleans his lab. He created the Animal Detector, a motion-sensitive webcam that films critters that wander into Goldstein’s suburban back yard to eat the food that he and his young son Duncan leave out for them at night (tally so far: raccoon, squirrel, opossum, rabbit, cat, robin, sparrow, wren, cardinal, gray fox, earthworm, human). He has biology-lab gadgets, as well: two fancy microscopes that can record multiple optical planes over time. Goldstein uses these microscopes to create videos of tardigrade embryos developing. “You can play back these films and see cells dividing on one side of the embryo and cells dividing on the other side,” he says. “And from that, you can reconstruct cell lineages: you can watch from the one-cell stage as it divides to two cells, and then four, and so on. And you can figure out the relationships between the cells, which can be important for just broadly understanding how development works.”
Goldstein and Tenlen are looking closely at germ cells in tardigrade embryos. Germ cells, which are made by all sexually reproducing animals, are sex cells: as an embryo develops, it sets aside one cell to become a germ cell, and this cell will eventually become either an egg or a sperm. In a sense, Goldstein says, the germ cell carries the organism’s immortality: it’s the germ cell’s DNA that is passed on when an organism reproduces. But we don’t really know a lot about how organisms single out a specific embryonic cell to become the germ cell. Goldstein and Tenlen think that in the tardigrade, two embryonic cells may compete for the honor. If that turns out to be true, and if Goldstein and Tenlen can figure out exactly how and why it happens, they may have taken a step toward figuring out how development influences evolution.
“Evolution is the topic that unites all of biology,” Goldstein says. “In my mind, development is the part of evolution that we’ve been missing for a long time.”
Bob Goldstein is an associate professor of biology in the College of Arts and Sciences. The National Science Foundation funds Goldstein’s tardigrade work. See Goldstein’s videos and images of tardigrades at http://tardigrades.bio.unc.edu/. Goldstein’s Animal Detector appeared in the March 2009 issue of Make magazine. In October 2009, a Russian spacecraft will take tardigrades to Phobos, one of the moons of Mars, to see if they can survive a three-year space journey.