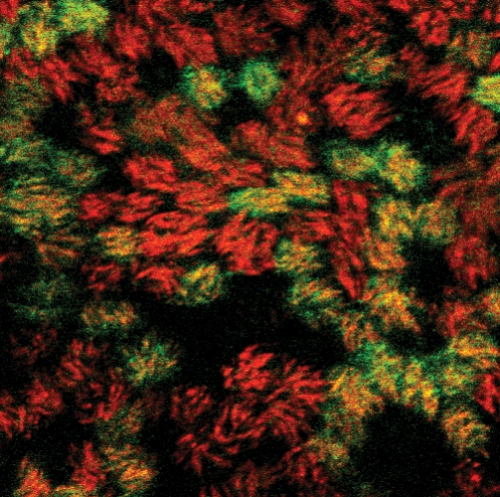
Just kind of stare at one spot,” Meg Scull said as she adjusted the focus. I peered through the lenses and saw yellow cells assembled in a cobblestone pattern.
It took a moment, but then I could see it — the cells were twitching in unison, making the liquid on top of them swirl like a whirlpool. I was watching the lining of a windpipe — human airway epithelium (HAE) — living and growing in a plastic dish.
“Sometimes when I need a break, I’ll just come in here and watch them,” Scull said with a laugh. Scull, a microbiology and immunology grad student working with Ray Pickles, is using the HAE model to figure out why bird flu isn’t spreading among humans.
The HAE model twitches because the top layer of cells is ciliated. The hair-like structures sway together rapidly, carrying foreign particles up and out of the lung like crowd surfers at a rock concert. But because the model HAE grows in a dish, the cilia just move things in a circle, creating the whirlpool.
The ciliated epithelium cells of the HAE are where an inhaled flu virus replicates if an infection is successful. If bird flu infects someone’s HAE, it could spread from person to person around the globe.
Bird flu migration
The H5N1 strain of bird flu has been running rampant in wild fowl and devastating poultry populations in Asia for years, and has slipped in and out of media attention in the process. In 2004, Time Asia Magazine speculated that H5N1 could be the next human pandemic. “H5N1 is making big news again because they’ve had three deaths so far in 2009 in China,” Pickles says.
Since 2004, H5N1 has spread to avian populations in Europe and Africa. There have been relatively few cases of H5N1 infection in humans, but when the virus does infect a person it tends to be deadly.
“All influenza viruses have originally come out of avian species,” Pickles says. “H5N1 is just one example of an avian flu that can get into the population with the possibility of a pandemic.”
Waterfowl carry influenza viruses in their GI tracts with few or no symptoms and constantly shed viruses. So places such as ponds where waterfowl often congregate are swarming with bird flu viruses, Pickles says. “Why aren’t we just picking these viruses up willy-nilly?”
When influenza H5N1 reaches chickens, they do get infected — and something strange happens. There’s an actual change in the virus, allowing it to infect all kinds of cells, not just those found in the bird’s GI tract. “In chickens, no organ is spared and the virus becomes rampant,” Pickles says. This is why poultry workers have made up the majority of cases of H5N1 in humans.
“Because of the sheer amount of virus that’s being produced, you have the susceptibility of the poultry workers to get inoculated with high doses,” Pickles says. Poultry workers handling infected chickens would be exposed to infected tissues, blood, and aerosols, all containing extremely high levels of flu virus. “That’s a very different exposure than you see with somebody sneezing on a table and you picking it up two days later,” he adds.
Scientists and public-health experts studying the flu have accepted the idea that an unusually high level of H5N1 is necessary for it to make the jump from birds to humans. But are we closer to understanding the risk to public health?
“The big question now is the whole transmission thing,” Pickles says. If the virus could be transmitted, it could move from the few infected poultry workers to people around the world.
Enter the flu
Once a flu virus enters the human airway, it needs to usurp a cell’s machinery and replicate. The ciliated cells of the airway’s epithelium are easy targets.
To avoid being carried away by the swaying cilia, the flu virus uses surface proteins to attach and drag itself into the cell. These proteins, called hemagglutinin and neuraminidase, are found all over the surface of the viral particle.
Hemagglutinin and neuraminidase have been shown to be crucial for flu infection. In fact, the designation for flu viruses refers to the type of hemagglutinin and neuraminidase the virus carries: H5N1 carries the fifth defined type of hemagglutinin and the first defined type of neuraminidase. Most vaccines against human flu target hemagglutinin. The reason we get vaccinated each year is because hemagglutinin continually mutates.
Hemagglutinin binds to a sugar called sialic acid on the ciliated airway epithelial cells. Once the binding occurs, the other viral surface protein, neuraminidase, cuts the two apart. In essence, the viral particle claws its way to the cell by continually binding and releasing sialic acid with hemagglutinin and neuraminidase, like a retractable grappling hook. “These two sort of evolved together to match the activity of the other,” Scull says.
When the virus makes it to the cell, it enters and starts the infection process. “The virus ends up replicating in the nucleus and then gets transported back out and assembled,” Scull explains.
When the new viral particles are assembled and ready to leave the cell, the hemagglutinin and neuraminidase allow the virus to pull its way up and out so that it can infect the next cell.
The transmission thing
“When we get a circulating flu virus, we’re coughing and sneezing, depositing stuff everywhere; people are picking it up,” Pickles says. If you’re infected, the expanding viral population and the subsequent immune response damage your airway epithelium, which causes coughing and sneezing as your body tries to clear the debris.
“But with the documented cases of H5N1, it seems to be a different story,” Pickles says. “It seems that there are very few common cold symptoms. There’s no sneezing and most of the infection seems to be taking place in the lower airways and the lungs, so it’s more like pneumonia.”
If you don’t sneeze and cough, it’s much harder to transmit the virus.
Scull and Pickles think there are at least two reasons why H5N1 doesn’t transmit well in humans. The first is that the hemagglutinin and neuraminidase of H5N1 have adapted to function best at the temperature of the avian GI tract, which is much warmer than our upper airways. The other reason has to do with the type of sialic acid the hemagglutinin binds to.
The temperature of the avian GI tract is around 104 degrees Fahrenheit, while the human upper airway epithelium, where human flu viruses infect, is around 89 degrees. Scull and Pickles’ data show that the bird flu hemagglutinin and neuraminidase are most efficient at 104 degrees.
They took an attenuated human flu virus and engineered it to carry hemagglutinin and neuraminidase from avian flu. Then they infected their model HAE with this avianized human flu. By measuring the level of virus in the HAE, they found the avianized human flu was much worse at infecting when the HAE was at 89 degrees — the temperature of the upper airways.
“It’s not a huge molecular structure that people spend years studying,” Pickles says. “It’s related to the simple fact that we have a different temperature in our airways. It’s an innate defense.”
But our lower airway epithelium is 98 degrees, closer to the temperature of the avian GI tract. If a person were exposed to high levels of H5N1 — such as the exposure levels poultry workers face — infection might occur in the lower lung.
“And if I were to guess, I would say that once the virus gets into that lower lung, it’s really, really hard to control,” Pickles says. Our immune system controls viral infections with inflammation. “Sending inflammatory cells into the lower lungs has a much greater consequence than sending them into our big airways where we can clear things.”
Not only is the avian hemagglutinin adapted to function at higher temperatures, it also preferentially binds to different sialic acids.
Sialic acid is a generic sugar that can attach to other sugars in at least two ways: 2,3 and 2,6 linkages. The hemagglutinin of avian flu viruses preferentially binds to sialic acids that are in a 2,3 linkage, meaning that the second carbon on sialic acid attaches to the third carbon of another sugar. Hemagglutinin of human flu viruses, on the other hand, tends to bind to sialic acids that are attached to other sugars in a 2,6 linkage.
“In influenza pandemics, all of those viruses bound to 2,6 linked sialic acid,” Pickles says. “So it seems to be that if you’re a flu virus and you want to go from person to person, you need to bind to 2,6 linked sialic acid. We don’t really know what advantage that confers to allow for transmission between people.”
The lab’s data suggest that if you have a virus that prefers 2,6 linked sialic acid, it does better at the temperature of the upper airway epithelium. “So it could be that mutating to prefer 2,6 linked sialic acid enables you to replicate at a lower temperature,” Scull says.
If H5N1 were to mutate to infect the upper airway epithelium like seasonal human flu viruses do, would it be less likely to enter the lower lung and cause life-threatening disease? Or would it just be a transmissible virus that our immune systems won’t recognize?
“Flu is a virus that’s so common because it has this incredible ability to mutate. And although small mutations are actually okay for us, because antibodies give us some protection, big mutations — such as when you introduce a new hemagglutinin into the population — have the potential to be devastating,” Pickles says. “When we first got into influenza I thought, ‘What is there to do in influenza research?’ because this has been studied for years. It’s really amazing what we still don’t know.”
Beth Mole was formerly a postdoctoral fellow in the medicinal chemistry and natural products division of the Eshelman School of Pharmacy.
Beth Mole is a doctoral student in the Department of Microbiology and Immunology in the School of Medicine.Ray Pickles is an associate professor in the Department of Microbiology and Immunology. Funding for his bird flu research comes from the National Institutes of Health.